Measurement of the intracranial pressure during space flight is critical to understand whether the visual impairment intracranial pressure (VIIP) syndrome is related to pathological elevation of the ICP. Currently only invasive measurement of ICP is considered accurate enough for determination of this parameter. Given that all invasive methods for measuring ICP carry some risk, this may be catastrophic in the space flight environment. Therefore development of non-invasive technologies for measuring the astronauts’ ICP is highly desirable. This study aims to evaluate the latest generation of a transcranial Doppler based technology for measuring ICP against gold standard invasive methods in patients on Earth requiring ICP measurement. The results of this study will help determine the accuracy and usability of this latest version of the non-invasive ICP meter (Vittamed).
Overview
Clinical Evaluation of the Newest Generation of a Non-Invasive ICP Meter (VITTAMED) in Subjects Undergoing Invasive ICP Measurement
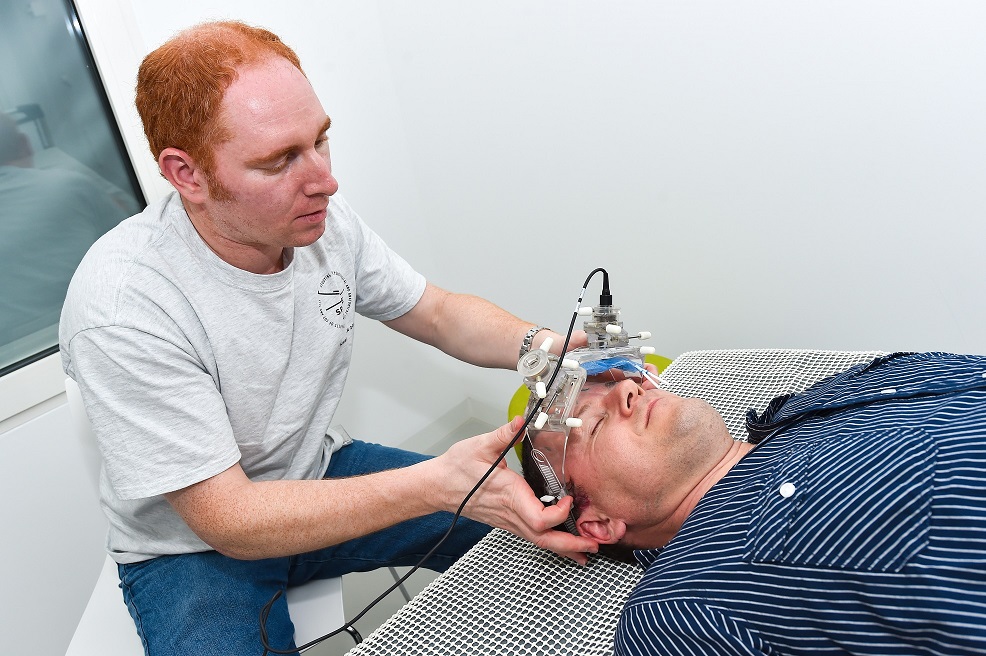
Dr. Bershad works with a previous version of the Vittamed device.
Principal Investigator:
Eric Bershad, M.D.
Organization:
Baylor College of Medicine
Technical Summary
The study will enroll 60 patients undergoing ICP measurement either by lumbar puncture or intracranial CSF pressure (Ommaya reservoir). The Vittamed device uses ultrasound to measure two different segments of the ophthalmic (eye) artery (OA) at the same time. Differences between the flow through the eye artery are analyzed to assess the effect of the ICP on the intracranial segment as compared to the extracranial arterial segment which does not have ICP pushing on the arterial wall. An external pressure is applied via incremental inflation of a soft orbital cuff through a range of pressure. The external pressure applied at which the flow velocity pattern appears most similar between the intra- and extracranial OA segments determines the ICP.
The primary aim of the study is assessing the accuracy of the non-invasive method compared to the simultaneously measured invasive ICP measurement. The secondary aim is to assess the change in ICP related to either CSF drainage (lumbar puncture) or body tilt (upright / supine) in ommaya reservoir patients. Study sites will include Baylor College of Medicine, UT Dallas Southwestern, and MD Anderson. The results of this study will help guide NASA and the NSBRI as to whether this device is accurate enough for measurement of the astronauts’ ICP before, during and after space flight.
Earth Applications
Many patients with brain injury from various causes require measurement of ICP to improve long-term neurological function, and survival. This technology if accurate would be valuable for use in settings such as the neurocritical care unit, emergency room, battlefield, high altitude, and remote settings, where invasive measurement of ICP may be difficult due to risks, lack of expertise, or costs. Since a non-invasive device that can accurately measure ICP does not currently exist, development of this and other devices having potential for accurate measurement of ICP is very important.





